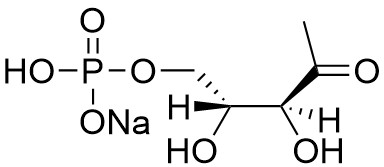
Isoprenoid compounds are a diverse group of natural products which are essential components in all cells. Isoprenoids are biosynthesized from the simple precursors isopentenyl diphosphate (IPP) and dimethylallyl diphosphate (DMAPP). Eukaryotes, fungi, and some gram-positive bacteria produce IPP through the mevalonate (MVA) pathway whereas gram-negative and some gram-positive bacteria utilize the non-mevalonate or 2-C-methyl-D-erythritol-4-phosphate (MEP) pathway. 1-Deoxy-D-xylulose 5-phosphate (DXP) is an intermediate in the non-mevalonate pathway and is formed from pyruvate and glyceraldehyde 3-phosphate by DXP synthase. It is converted to MEP by MEP Synthase (DXP reductoisomerase).
Provided as the sodium salt