Isoprenoids are an amazingly diverse and biologically important class of molecules. They are some of the oldest known biomolecules, with evidence that one class of isoprenoids, the hopanoids, have existed for at least 2.5 billion years. Isoprenoids can be categorized by the number of isoprene units which have been linked together, from one isoprene unit (hemiterpenoid) to eight or more isoprene subunits (polyterpenoids). They can also be modified in an almost unlimited manner, giving rise to many well-known and essential classes of molecules. For example, the carotenoids, a ubiquitous class of isoprenoids in fungi and plants, are synthesized from geranylgeranyl diphosphate, and are pigments involved in photosynthesis and signaling. Additionally, sterols are cyclized compounds derived from isoprenoids that play roles in cell membrane rigidity and hormonal signaling. Below we will examine additional details of isoprenoid function, synthesis, and their potential use as bioactive compounds & drugs.
Isoprenoid structure and biosynthesis
Isoprenoids, also known as terpenoids, consist of a backbone of repeating isoprene monomers. However, they are actually formed in vivo from the precursors isopentenyl diphosphate (IPP) and dimethyallyl diphosphate (DMAPP) shown in Figure 1. IPP and DMAPP are acted upon by a number of enzymes, with a major route of isoprenoid synthesis combining the above precursors via geranyl pyrophosphate synthase to form geranyl diphosphate (GPP), which is then processed through various intermediates to form over 30,000 known examples of isoprenoids.
IPP and DMAPP are themselves formed via the mevalonate (MVA) pathway in eukaryotes, archea, and some eubacteria and the 2-C-Methyl-D-erythritol 4-phosphate (MEP) pathway in bacteria, plastid containing eukaryotes and some protazoans. The mevalonate pathway has four shared synthesis steps in eukaryotes and archea that synthesize mevalonate 5-phosphate from acetyl-CoA. The remaining steps that ultimately produce IPP and DMAPP diverge in terms of the enzymes and intermediates that are utilized. The MEP pathway on the other hand uses a completely different synthesis path than the MVA pathway.
The MEP pathway begins with the condensation of pyruvate and glyceraldehyde 3-phosphate. The resulting product, deoxyxylulose 5-phosphate is then used to produce MEP and represents the first committed step of the pathway. This and all subsequent steps that ultimately produce IPP are performed by a series of enzymes IspC-F and are unique to this pathway. Because the MEP pathway is largely confined to bacteria, it has drawn intense interest as a potential target for novel classes of antibiotics.
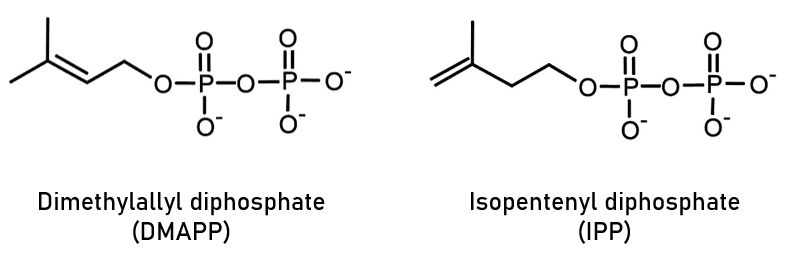
Figure 1: Chemical structures of the two major isoprenoid precursors.
Modern drugs and bioactive compounds
The rise of multi-drug resistant and antibiotic resistant pathogens has led to research into drugs with novel mechanisms of action. The MEP pathway represents an attractive set of targets as each enzyme is potentially druggable. A leading example is the antibiotic fosmidomycin, an inhibitor of IspC (DXP reductoisomerase). Fosmidomycin was initially characterized as an antimalarial agent and has since been studied as a general antibiotic. Subsequent work showed that it was also active against gram-negative bacteria. Multiple derivatives of fosmidomycin have since been produced with goals of improved bioavailability and efficacy against predicted IspC mutations that may confer resistance.
Many important drugs and bioactive compounds are also in the isoprenoid class, including the anticancer drug paclitaxel, which is synthesized from isoprenoid precursors, as well as nitrogen containing molecules, making it both an isoprenoid and an alkaloid. Salvinorin A, a natural isoprenoid synthesized in salvia divinorum, is a kappa opioid receptor agonist, and consuming it can lead to dissociative experiences. The cannabinoid class of molecules are produced both endogenously in animals (endo cannabinoids) as well as by plants with psychoactive effects when ingested by animals. Menthol, ginkolides, steviol, camphor, curcuminoids, and even natural rubber are all examples of isoprenoid compounds produced by plants that have important biological and practical applications in use today. Endogenous proteins can be isoprenylated , which is generally thought to increase their ability to associate with membranes and effect subcellular targeting. Figure 2 shows some examples of the wide range of chemical structures in the isoprenoid class of molecules.
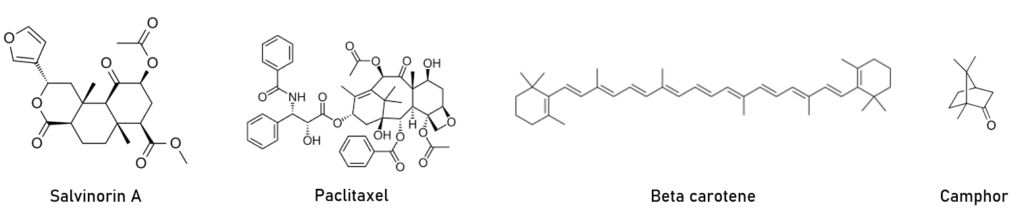
Figure 2: Examples of natural and synthetic compounds derived from isoprenoid precursors.
Outlook
The field of isoprenoid metabolism continues to identify new IPP-derivatives across disparate areas such as plant physiology to immunology. Similarly, the discovery of novel MEP class antibiotics is an ongoing area of intense research. Given the broad utility of isoprenoids and the current need for novel antibiotics, Echelon Biosciences will continue to invest in and support these research areas. We currently offer many simple isoprenoid compounds and biosynthetic intermediates, which can be used in both biological experimentation and as precursors in chemical syntheses. We have also developed a series of assays for activity of the MEP enzymes which we hope will aid in the development of novel compounds that can target drug-resistant pathogens.
View all Isoprenoid Products View MEP Pathway Screening ServiceReferences
1. Odom, A (2011) Five Questions about Non-Mevalonate Isoprenoid Biosynthesis. PLOS Pathogens 7 (12)
2. Masini T, Hirsch A.K.H.(2014) Development of Inhibitors of the 2C-Methyl-d-erythritol 4-Phosphate (MEP) Pathway Enzymes as Potential Anti-Infective Agents. J Med Chem 57, 9740−9763
3. Lombard J, Moreira D (2011) Origins and Early Evolution of the Mevalonate Pathway of Isoprenoid Biosynthesis in the Three Domains of Life. Molecular Biology and Evolution, 28 (1) 87–99
0.2
/ 0.3
Related Articles
Stay informed with our informative blog posts.
0.3
/ 0.3
Get in Touch
If you have any questions or would like to learn more about our services, feel free to reach out to us. We’re here to help!
Biosciences