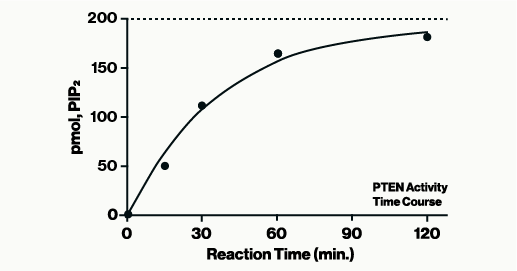
The PTEN Activity ELISA is designed to quantify the phosphatase activity of PTEN by detection of the enzyme product, PI(4,5)P2.
Sample Type: Recombinant or immunoprecipitated PTEN enzyme (IP protocol at end of Technical Data Sheet)
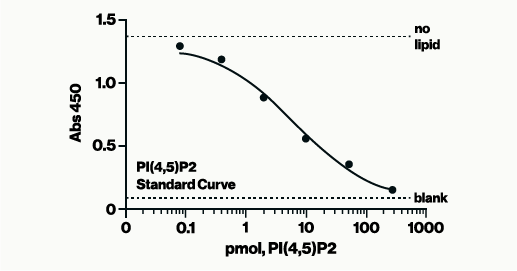
Assay Range: 12.5 to 200 pmol PI(4,5)P2
Product Background
The PTEN Activity ELISA directly detects the phosphoinositide product. Other assays detect free phosphate. This eliminates many possible sources of error due to the fact that inorganic phosphate is the product of many phosphatase enzyme activities, and is found in common buffers and cleaning products.
The assay is a competitive ELISA in which the signal is inversely proportional to the amount of PI(4,5)P2 produced. After the PTEN reactions are complete, reaction products are added to the PI(4,5)P2-coated microplate and a PI(4,5)P2 detector protein is added for competitive binding. A peroxidase-linked secondary detector and colorimetric detection is used to detect PI(4,5)P2 detector binding to the plate. The colorimetric signal is inversely proportional to the amount of PI(4,5)P2 produced by PTEN.
PTEN (Phosphatase and Tensin Homolog deleted on Chromosome 10) is a 3’ phosphoinositide phosphatase that converts PI(3,4,5)P3 to PI(4,5)P2, thus opposing PKB/Akt activation by PI 3-K. PTEN is involved in neuronal stem cell proliferation and self-renewal, cardiac myocyte hypertrophy and contractility, and a wide range of developmental processes. PTEN, however, is best known for its role as a tumor suppressor. Loss of PTEN activity results in accumulation of PI(3,4,5)P3, abnormal activation of PKB/Akt, unregulated cell growth, suppression of apoptosis, and increased tumorigenesis in a number of human tissues. It has also been proposed that PTEN is a candidate for targeted chemotherapy because certain anti-cancer agents preferentially destroy tumors with PTEN mutations. In addition to this direct role in cancer, PTEN has recently been shown to regulate cancer-associated pathways including VEGF-mediated angiogenesis and others.