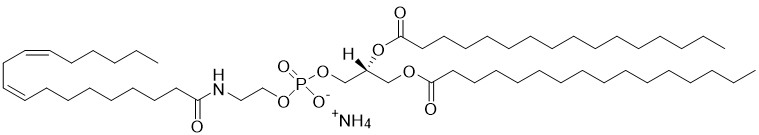
1,2-dipalmitoyl-sn-glycero-3-phosphoethanolamine-N-linoleoyl (N-linoleoyl-DPPE) is a specific non-endocannabinoid type of N-acylphosphatidylethanolamine (NAPE). NAPEs are the substrates of a specific phospholiase D (NAPE-PLD) which produces aqueous-soluble bioactive fatty acid amides or N-acylethanolamines. Linoleoylethanolamide is a non-endocannabinoid N-acylethanolamine and are more abundant as a group than anandamide and other endocannabinoid (which bind CB1 and CB2) in most tissues. The non-endocannabinoid group also have higher kD values for fatty acid amide hydrolases (FAAH) the enzyme that shuts off the bioactive signals by metabolizing N-acylethanolamines (NAEs). N-linoleoyl-ethanolamine (LEA) is one of the two most abundant NEA found in chocolate which do not activate brain cannabinoid receptors but effectively inhibit anandamide degradation by plugging up FAAH. More recently, LEA was found to protect neurons in the retinal ganglion cell layer against excitotoxicity and could be involved in type 2 diabetes as LEA is one of several ligands that bind GPR119.
L-2420
N-Linoleoyl-DPPE
1,2-dipalmitoyl-sn-glycero-3-phosphoethanolamine-N-linoleoyl (N-linoleoyl-DPPE) is a specific non-endocannabinoid type of N-acylphosphatidylethanolamine (NAPE). [Read More...]