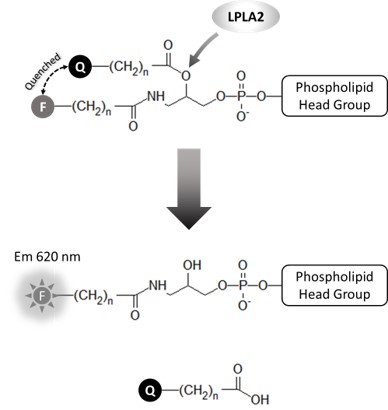
Echelon’s Lysosomal Phospholipase A2 (LPLA2) Activity Assay is a homogenous assay designed to detect activity from biological and purified sources of LPLA2. This in vitro, plate-based assay uses a quenched substrate which fluoresces when hydrolyzed by LPLA2. The sn-1 position of the quenched substrate is protected by an amine group to eliminate non-specific turnover by phospholipase A1. The acidic assay conditions further improve selectivity of LPLA2 for hydrolysis of the quenched substrate. This direct biochemical approach provides a specific, quantitative measurement in a robust and easy-to-use format for measurement of LPLA2 activity.
Product Background
Human LPLA2, also known as Lp-PLA2 or PLA2G15, is an enzyme responsible for normal lipid metabolism and is unique from other known PLA2s. LPLA2 is only active in acidic environments and research suggests it is related to drug-induced phospholipidosis (DIPL). DIPL is a condition of excessive accumulation of intracellular phospholipids triggered by long-term use of certain cationic amphiphilic drugs (CADs) such as fluoxetine (Prozac, Sarafem) and Amiodarone. DIPL results in tissue inflammation and organ damage. If not treated, DIPL can be fatal. The FDA has determined that DIPL is a serious drug safety issue.
Storage: -20°C. Avoid frequent freeze-thaw cycles.