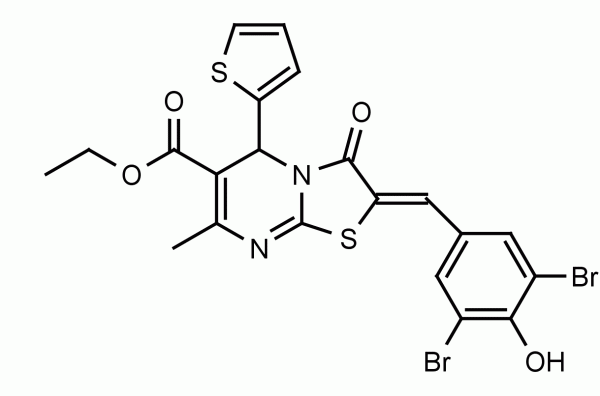
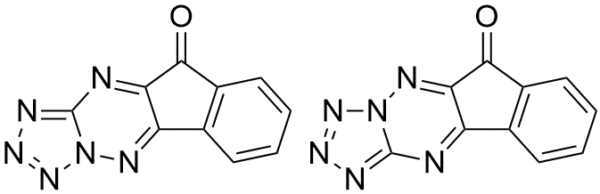
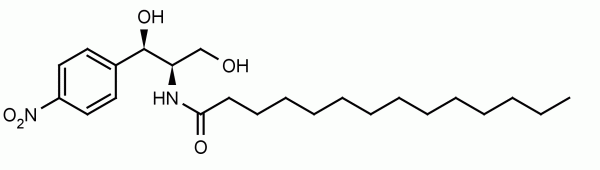
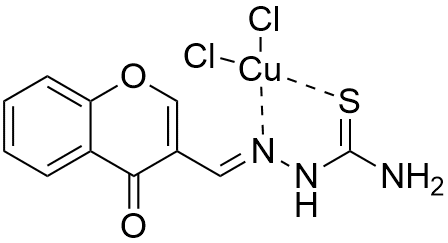
The non-mevalonate, or MEP, pathway for isoprenoid biosynthesis is essential in gram-negative, some gram-positive bacteria, plants, and protozoa making it an attractive pathway to target for antibiotics since mammals use the mevalonate pathway to synthesize isoprenoids. IspF is the fourth step in the pathway and converts diphosphocytidyl-2-methylerythritol 2-phosphate into 2-methylerythritol 2,4-cyclodiphosphate (cMEPP). IspF inhibitor 1 (compound 3 in Geist et al.) has low micromolar activity (IC50 = 6.1-32 μM) against the recombinant enzymes and inhibits the growth of P. falciparum in a red blood cell assay.
Product Number: B-4102
B-4102
IspF Inhibitor 1
The non-mevalonate, or MEP, pathway for isoprenoid biosynthesis is essential in gram-negative, some gram-positive bacteria, plants, and protozoa making it an attractive pathway to target for antibiotics since mammals use the mevalonate pathway to synthesize isoprenoids. [Read More...]