In mammals, the well-being of offspring can be influenced by the maternal diet and environment. However, it is not clear how these factors affect cellular functions across multiple generations. Previous work has described how intergenerational inheritance uses epigenetic mechanisms that involve RNAs and DNA modifications. Whether these mechanisms extend to utilize non-nucleic acid molecules such as proteins, lipids, or small metabolites has not been well explored. In the current study, researchers discovered that feeding Caenorhabditis elegans ursolic acid, a natural substance found in plants, had positive effects on axon transport and enhanced stability of axons in subsequent generations of worms.
This neuroprotective effect was mediated by enhancing the maternal provisioning of sphingosine 1-phosphate (S1P), a bioactive lipid, to offspring. The transfer of S1P from the intestine to oocytes (egg cells) was crucial for this intergenerational neuroprotection and depended on a specific lipoprotein yolk receptor called RME-2. S1P, in turn, played a role in increasing the expression of the acid ceramidase-1 (asah-1) gene in the intestine, affecting sphingolipid metabolism. The neuroprotective impact of intestinal asah-1 relies on the presence of SphK-1, an enzyme responsible for converting sphingosine into S1P. In this context, S1P supplied by the mother is transferred from the intestine to the oocytes to support axons. Importantly, this may be a conserved mechanism as S1P and SphK have also been found to play vital roles in protecting vertebrate oocytes against apoptosis.
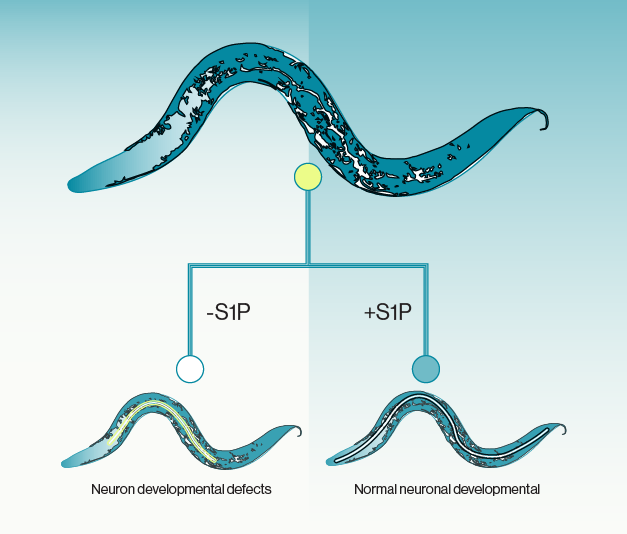
Periods of interaction between mothers and offspring enable maternal influence on subsequent generations. Changes in the reproductive behavior of offspring can facilitate the transmission of these effects to succeeding generations. This study provides evidence that the transmission of a lipid metabolite within the yolk can provide intergenerational protection to specific neurons. These findings highlight the significance that specific lipid molecules can transmit information between generations which may have broad implications for regulating metabolism during reproduction. Echelon’s S1P Fluorescein (S-200F) was used in this work.
Read the full article here:
An intestinal sphingolipid confers intergenerational neuroprotection
Nature Cell Biology 25:1196-1207 (2023)
0.2
/ 0.3
Related Articles
Stay informed with our informative blog posts.
0.3
/ 0.3
Get in Touch
If you have any questions or would like to learn more about our services, feel free to reach out to us. We’re here to help!
Biosciences