Communication between neurons in the brain, neurotransmission, is dependent on the release of small molecules from specialized structures called synaptic vesicles (SVs). The release of these small molecule neurotransmitters from SVs is dependent on fusion of SVs with the neuronal cell membrane followed by SV recycling to be reloaded with neurotransmitters. This cycle of exo and endocytosis is critical for maintaining proper signaling homeostasis between neurons, although how these seemingly coupled events are balanced to sustain neuronal function has not been shown.
Here, Bolz & Kaempf and colleagues provide evidence that the calcium sensor Synaptotagmin 1 (Syt1) links SV exocytosis to endocytosis by recruiting a lipid kinase for local synthesis of the phosphoinositide PI(4,5)P2 at exocytic sites. Previous work has shown that PI(4,5)P2 can recruit adaptor proteins to the membrane for the assembly of clathrin-coated pits for clathrin mediated endocytosis. Using a mouse knock out model for Syt1, they show that re-expression of Syt1 restores local PI(4,5)P2 synthesis via interactions with the kinase PIPKIγ. Similarly, expression of a Syt1 mutant that lacks PIPKIγ binding only restores exocytic function, but endocytosis of SVs remains impaired.
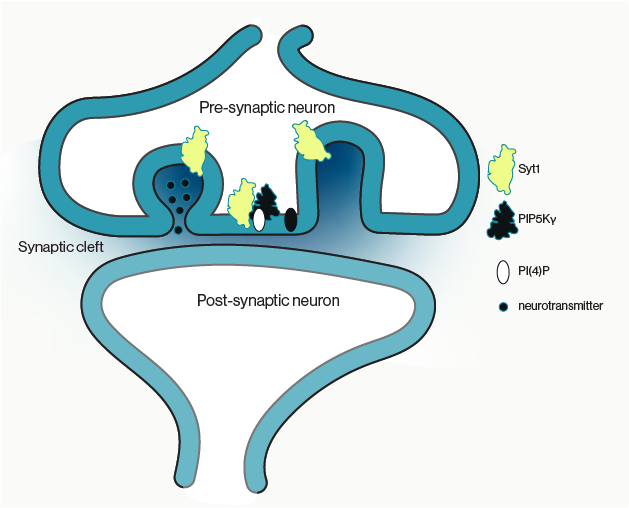
The current data provides evidence in support of a model where exo-endocytic coupling is dependent on local lipid signaling in the membrane. However, this does not exclusively rule out the possibility that some SV proteins that have been exocytosed are also required as adaptors for other endocytic proteins. The authors propose that Syt1 acts as both a platform and sensor for PI(4,5)P2 synthesis such that as PI(4,5)P2 levels rise PIPKIγ is released from the complex. This mechanism would allow for downstream recruitment of clathrin-mediated endocytic proteins such as the adaptor AP2, as well as freeing PIPKIγ for subsequent rounds of PI(4,5)P2 synthesis following exocystosis.
Read the full article here:
Synaptotagmin 1-triggered lipid signaling facilitates coupling of exo- and endocytosis
Neuron 111, 1–10, December 6, 2023; online September 21, 2023
0.2
/ 0.3
Related Articles
Stay informed with our informative blog posts.
0.3
/ 0.3
Get in Touch
If you have any questions or would like to learn more about our services, feel free to reach out to us. We’re here to help!
Biosciences