A new SBIR grant through the Department of Defense aims to develop new prophylactic treatments for exposure to chemical warfare agents (CWA) such as sarin. The program’s main goal is large-scale production of active butylcholinesterase (BChE), a naturally occurring enzyme in the blood that is known to be effective in humans against CWA exposure. The enzyme acts as a bioscavenger that binds CWAs in the bloodstream and prevents them from affecting target organs.
Paul Neilsen, PhD., Director of R&D at Echelon Biosciences, developed the Phase 1 aims in collaboration with Dr. Don Jarvis at The University of Wyoming and his start-up, GlycoBac.
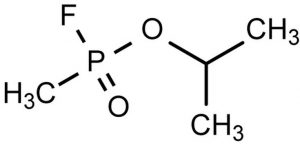
Figure 1: Structure of sarin gas, one of the most common organophosphate chemical warfare agents and one of the main targets of prophylactic BChE.
Recombinant BChE as a treatment
While BChE is known to block the effects of CWAs, there have been numerous obstacles to producing it in an active form at scale.The enzyme is maximally active as a tetramer and there are specific post-translational glycosylation and sialylation patterns required for the complex to be stable in the blood. Previous attempts to purify the complex from plasma or alternative expression systems have resulted in low yields, low activity, or improper post-translational modifications.
Dr. Jarvis’s group at GlycoBac has developed a unique, genetically engineered insect cell expression system that is capable of modifying proteins with human glycosylation and sialylation patterns. In parallel, the R&D group at Echelon has outlined novel purification techniques to capture recombinantly expressed BChE (rBChE) in its tetrameric form.
The goal of this collaborative effort is a system that overcomes the previous shortcomings to produce pure rBChE protein in large enough quantities to deliver an effective CWA treatment.
Looking ahead
While the importance of rBChE is obvious, producing proteins with human glycosylation and sialylation is a critical challenge in many areas. A prime example is the engineering of therapeutic antibodies.
Antibody therapy has grown rapidly in the last decade, and antibodies with the proper glyco modifications appear to have greater efficacy. The rBChE project will serve as a proof-of-principle for this glycoengineered protein-expression platform.
0.2
/ 0.3
Related Articles
Stay informed with our informative blog posts.
0.3
/ 0.3
Get in Touch
If you have any questions or would like to learn more about our services, feel free to reach out to us. We’re here to help!
Biosciences


