The PI4-Kinase Activity Assay has been designed to detect and quantify, the end product of a PI 4-Kinase reaction, PI(4)P.
Product Background
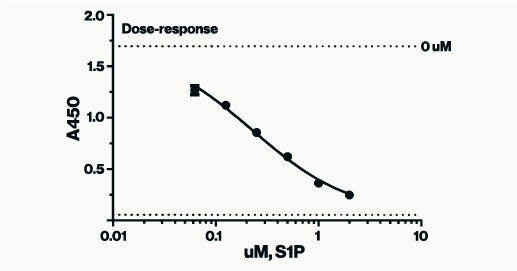
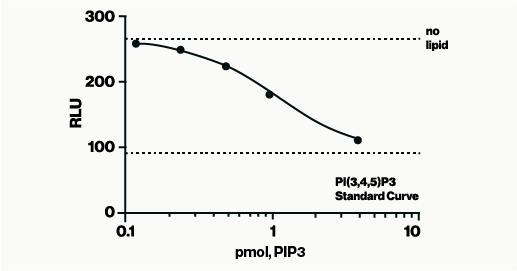
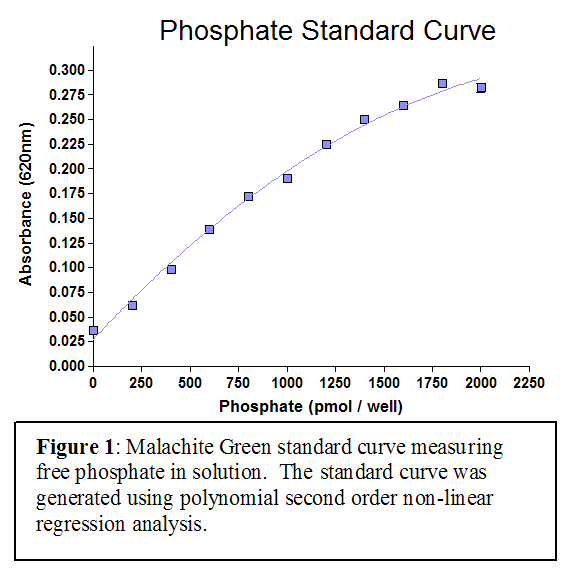
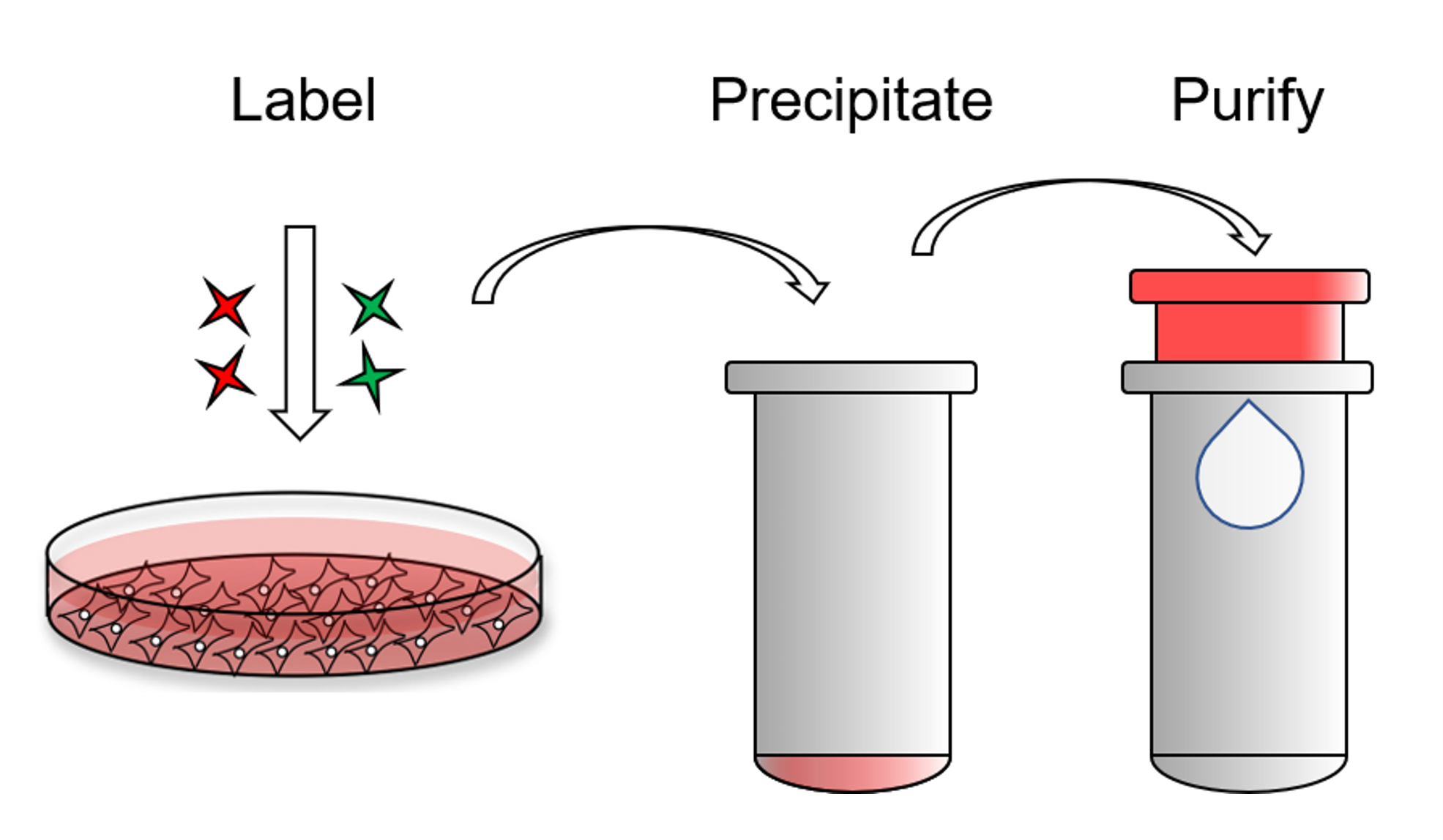
The PI4-Kinase Activity Assay is a competitive ELISA in which the signal is inversely proportional to the amount of PI(4)P measured. The enzyme reactions are stopped with EDTA before the PI(4)P Detector is added. This mixture is then transferred to the PI(4)P Detection Plate for competitive binding. A peroxidase-linked Secondary Detector and colorimetric detection is used to detect the amount of PI(4)P Detector binding to the plate.
Phosphatidylinositol 4-phosphate (PI(4)P) is the most abundant monophosphorylated phosphoinositide found in mammalian cells. It is produced by PtdIns 4-Kinases (PI4K) which phosphorylates the D-4 position of the inositol ring of PtdIns. PI 4-Kinases have been classified into two types, II and III, based on their molecular mass, and modulation by detergent and adenosine. Type II class PI 4-Kinases includes PI4KIIa (PI4K2A) which is involved in many biologic processes ranging from cell growth to endo- and exocytosis. It is also a novel regulator of tumor growth and generates PtdIns4P-rich domains within the Golgi. PI4KIIb (PI4K2B) is primary cytosolic where it is recruited to the membrane and stimulates PI(4,5)P2 synthesis. Type III PI 4-Kinase, PI4KIIIa (PI4K3A), plays a role in replication of the Hepatitis C virus. These viruses use PI4KIIIa to generate PI(4)P-enriched environments increasing cellular levels of PI(4)P. PI4KIIIb (PI4KB), regulates the golgi reorganization during mitosis. This enzyme is essential for glucose-induced insulin secretion due to its capacity to regulate the release of secretory granules. All of this makes PI 4-Kinases an interesting target for a diverse set of biological functions, disease states, and infections.