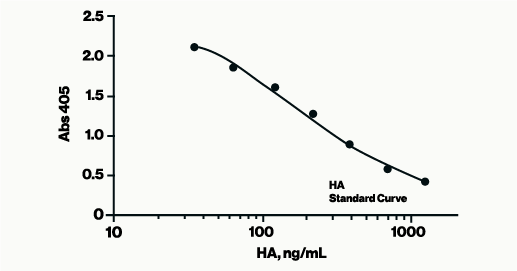
The Hyaluronan Enzyme-Linked Immunosorbent Assay (HA ELISA), a competitive 96-well assay, offers a simple, effective method for determining Hyaluronic Acid (HA) levels in human and animal biological fluids or cell supernatants.
Assay Range: 50-1600 ng/ml
Sensitivity: 25 ng/mL
Sample considerations: The HA ELISA detects HA molecules that are as small as 6.4 kDA.4
Hyaluronic acid (HA) is a high molecular weight anionic polysaccharide (1,000-10,000 kD) composed of repeating disaccharides. It is one of several glycosaminoglycan components of the extracellular matrix of connective tissue.
Free HA is taken up by the liver where it is degraded and recycled. Many chronic liver diseases, including infection (hepatitis B or C), toxicity (alcohol and drugs), genetic (hemochromatosis), autoimmunity, and malignancy, result in liver inflammation which can progress to liver fibrosis and cirrhosis; causing impairment of liver function and resulting in a rapid increase in circulating HA levels.
Data indicates a relationship between HA levels, local inflammation and severity of disease. Recent publications have shown HA levels in urine are indicative of bladder cancer, that HA levels are correlated to liver disease, and the breakdown of HA in the lungs of patients with chronic obstructive pulmonary disease. Additionally, serum levels of HA have been found to be elevated in patients with rheumatoid arthritis.
For help on selecting the correct HA detection assay see the HA Assay Comparison Chart.
For additional information on this product, see this Biocompare product review here and/or check out this article on “How to select Hyaluronic Acid (HA) quantitative tests?”
Publications
(see the HA ELISA TDS for more references)
1. Meuwese, M. C., H. L. Mooij, et al. (2009). “Partial recovery of the endothelial glycocalyx upon rosuvastatin therapy in patients with heterozygous familial hypercholesterolemia.” J Lipid Res 50(1): 148-153.
2. Vendrov, A. E., N. R. Madamanchi, et al. (2010). “NADPH Oxidases Regulate CD44 and Hyaluronic Acid Expression in Thrombin-treated Vascular Smooth Muscle Cells and in Atherosclerosis.” Journal of Biological Chemistry 285(34): 26545-26557.
3. B. KIM, et al. (2010). “Retinyl retinoate induces hyaluronan production and less irritation than other retinoids.” The Journal of Dermatology 37(5): 448-454.
4. Haserodt, S., M. Aytekin, et al. (2011). “A comparison of the sensitivity, specificity, and molecular weight accuracy of three different commercially available Hyaluronan ELISA-like assays.” Glycobiology 21(2): 175-183.
5. Hill, D. R., H. K. Rho, et al. (2013). “Human Milk Hyaluronan Enhances Innate Defense of the Intestinal Epithelium.” Journal of Biological Chemistry 288(40): 29090-29104.
6. Measurement of low-molecular weight HA: Schmaus, A, Klusmeier, S. et al (2014) “Accumulation of small hyaluronan oligosaccharides in tumour interstitial fluid correlates with lymphatic invasion and lymph node metastasisBr. J. Cancer 111(3): 559-567
7. Tang, S. C., S. J. Yeh, et al. (2014). “Association between plasma levels of hyaluronic acid and functional outcome in acute stroke patients.” J Neuroinflammation 11: 101.
8. Measuring HA mass distribution: Yuan, H., R. Amin, et al. (2015). “Determination of hyaluronan molecular mass distribution in human breast milk.” Analytical Biochemistry 474: 78-88.
Product Keywords: Hyaluronan, Hyaluronate, Liver Cirrhosis, Rheumatoid Arthritis, Hyaluronan, Osteoarthritis, Liver Fibrosis